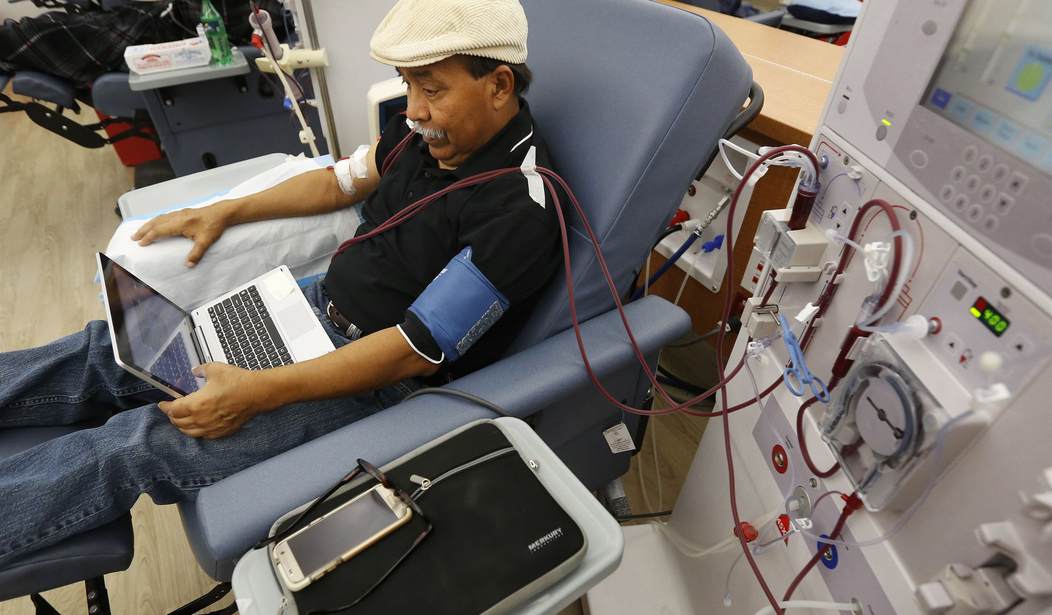
As many parts of the country begin to re-open, policymakers will face the challenge of how best to protect the vulnerable among us while ensuring Americans can safely get back to work. Among those most susceptible to the coronavirus include the more than 37 million Americans living with chronic kidney disease (CKD).
Even before COVID-19 became a global pandemic, the CKD patient community faced enormous challenges accessing the care they need. Now, as states prepare to re-open and get their economies going again, policymakers must ensure CKD patients — many of whom are immunocompromised — can continue to access medically-necessary care without risking viral infection.
This effort should start with removal of burdensome prior authorization (PA) requirements that prioritize bureaucratic process over the delivery of care. Long the bane of patients suffering from chronic conditions, prior authorization requires patients and their doctors to receive coverage approval from their insurance plan before proceeding with treatment. This means physicians, despite years of training and familiarity with the needs of their patients, must waste precious time and resources chasing down payers and getting signatures. Such requirements burden providers at a time when our healthcare system is already stretched thin and delay patient access to critical care.
To make matters worse, prior authorization regulations often fall heaviest on populations with chronic diseases such as CKD. This means the very patients most at risk of COVID-19 – those who are immunocompromised – must also deal with regulatory red tape before being able to access doctor-prescribed treatments.
Recommended
Maintaining such detrimental regulations, at least for the duration of this public health emergency, is terrible news for America’s kidney patients. Federal officials charged with overseeing the Medicare program have begun taking broad action to expand coverage and treatment access for vulnerable patient populations. Yet, while these steps are praiseworthy and significant, officials should go further and encourage health insurance companies to waive PA requirements until the worst of the COVID-19 pandemic is behind us. And federal officials should also ensure that vulnerable patients with diagnosed end-stage renal disease conditions (ESRD) – or kidney failure – should be able to access Medicare Advantage plans that can offer more robust and coordinated care options essential to vulnerable populations with multiple disease conditions by reinstating the enforceable network adequacy requirement that plans must include outpatient dialysis organizations in their networks.
Along with clearing away gratuitous bureaucratic coverage hurdles for CKD patients, another more long-term challenge that must be addressed is the potential for disease transmission among kidney patients in outpatient care facilities. Recent exposés by the Washington Post, New York Times, and other major publications have documented the deadly reality of viral spread in outpatient care settings. Much like dialysis patients, many CKD patients suffering from iron deficiency anemia (IDA) must still travel to outpatient facilities to receive the intravenous iron infusions. To mitigate the risk of viral exposure, federal officials should immediately expand access to an oral therapy that can treat IDA without the need for intravenous infusion.
The oral therapy, ferric citrate coordination complex, is an innovative compound that has been clinically proven to effectively treat iron deficiency anemia in CKD patients not on dialysis. Most importantly, the drug is taken orally – allowing patients to remain safely at home and avoid visits to outpatient facilities whose resources are already overburdened. First approved by the FDA in 2014, ferric citrate has been hailed by patients, physician groups, and leading CKD advocacy organizations like ours as a safe and cost-effective treatment for renal anemia.
But despite these clear advantages, the Centers for Medicare & Medicaid Services (CMS) – the federal agency responsible for administering Medicare – decided to significantly reduce access to this patient-preferred drug. In a September 2018 decision, CMS declared it would no longer provide reimbursement for ferric citrate to treat renal anemia under Medicare Part D. In other words, CKD patients were deprived access to the only FDA-approved oral treatment for iron deficiency anemia, forcing them in many cases to rely on other alternatives such as IV iron infusions.
In the midst of the present crisis, restoring coverage for oral treatments of renal anemia will allow many vulnerable CKD patients to maintain social distancing, mitigate their risk of contracting COVID-19, and free up hospital space to treat severely-ill coronavirus patients. Expanded coverage of oral-treatments delivered in home setting, in conjunction with broader-based efforts to clear away prior authorization requirements that undermine access to medically-necessary care, are important steps to support these vulnerable populations as the rest of the country starts to recover from the effects of this pandemic.
A third example of burdensome regulatory restrictions potentially harming patients during the pandemic is the decision not to clarify that Medicare beneficiaries who are COVID-19 positive or suspected of having the virus and require medical isolation can receive ground ambulance transport under the Medicare program. Not every patient should be transported by ambulance, but for those limited number who need the special medical isolation, it is important to have the access to these services. Without it, they may not be able to access their life-saving dialysis treatments, leading to fluid overload, cardiac issues, and likely hospitalization. CMS could fix this problem by instructing the contractors who pay claims to allow these transports during the public health emergency.
The 2020 COVID-19 pandemic has dramatically shifted the treatment paradigms of so many chronic conditions, including kidney disease. It has shed a light on ineffective systems and practices associated with CMS and the sometimes obsolete rules and guidelines currently in place. Let’s use this pandemic as an opportunity to examine ways to improve patient access and outcomes within the kidney community. Yes, COVID-19 has changed our world forever, but CMS should seize this opportunity to make sensible changes in kidney care to protect patients as they navigate a new normal.
























Join the conversation as a VIP Member