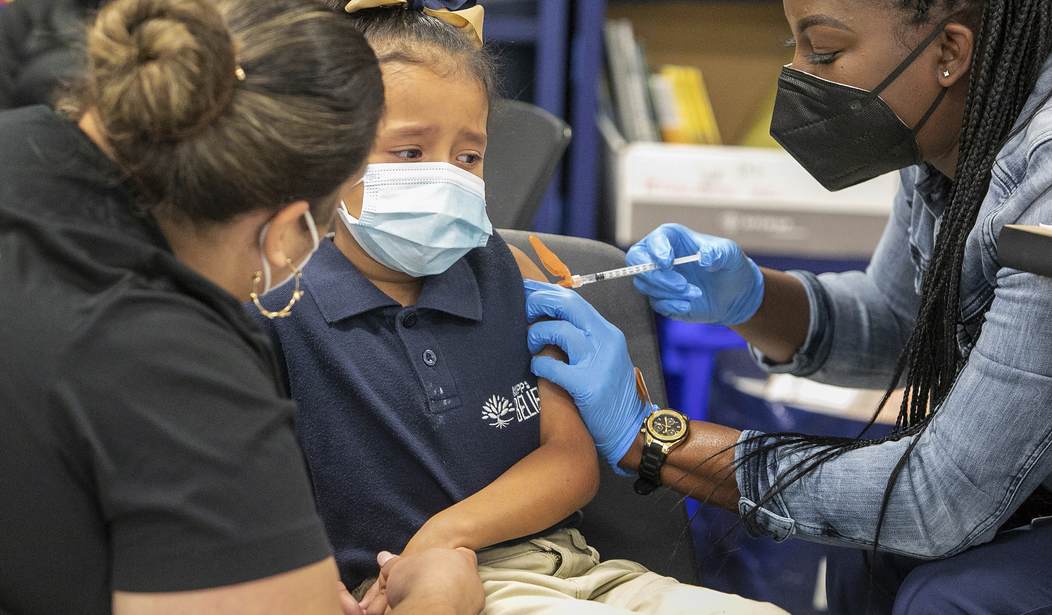
On Tuesday, the U.S. Food and Drug Administration (FDA) authorized a booster dose of the Pfizer-BioNTech COVID-19 vaccine for children ages 5 to 11.
The FDA published a news release on Tuesday explaining how the single-use booster can be administered to children in this age group at least five months after their primary two-dose vaccine.
“Since authorizing the vaccine for children down to 5 years of age in October 2021, emerging data suggest that vaccine effectiveness against COVID-19 wanes after the second dose of the vaccine in all authorized populations. The FDA has determined that the known and potential benefits of a single booster dose of the Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age at least five months after completing a primary series outweigh its known and potential risks and that a booster dose can help provide continued protection against COVID-19 in this and older age groups,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, in the release.
Townhall covered in December that the FDA authorized the Pfizer booster for 16 and 17 year olds as the Delta variant spread and the Omicron variant emerged. In early January, the FDA authorized a booster shot for children 12 through 15 years of age.
The release noted that the booster dose for 5 to 11 year olds was assessed in about 400 children five to nine months after they received their primary vaccine dosage. The most common side effects were pain, redness, swelling, fatigue, headache, muscle and joint pains, chills and fever.
Recommended
This age demographic is the youngest to date to receive authorization from the FDA for a COVID-19 booster shot. In February, Townhall reported how Pfizer and BioNTech were testing a third dose of the vaccine, following “disappointing” results from the two-shot regime in children under 5. Shortly after, Pfizer announced it was delaying the application process for Emergency Use Authorization for their vaccine for children ages 6 months through 4 years.























Join the conversation as a VIP Member