James Hildreth, a top vaccine adviser for the Food and Drug Administration (FDA), said the federal agency will decide on Thursday whether or not it will grant emergency use authorization (EUA) for Pfizer's coronavirus vaccine candidate.
"We'll spend the day on Thursday reviewing the data from Pfizer, and at the end of the day, a vote will be taken. So, by the end of the day next Thursday, there could be a decision made about the vaccine," Hildreth told NBC News on Saturday.
As far as when the first vaccinations might happen in the United States, the FDA adviser said that could begin as soon as Friday.
“If the FDA commissioner decides to issue approval, the EUA, on that day when the vote is taken, as early as Friday of next week, we could see vaccinations happening across the country," Hildreth added.
The Trump administration has prioritized the development of vaccines for the Wuhan coronavirus and news about Pfizer's development of a safe and effective vaccine candidate arrived much sooner than many top experts had predicted. And vaccinations beginning on Friday is more welcome news as coronavirus cases surge across the nation and some states and local governments prepare for another round of lockdowns.
Pfizer announced in early November that its vaccine candidate developed with BioNTech SE was show to be more than 90 percent effective in preventing COVID-19 infections, well above the FDA's efficacy requirement of 50 percent for a vaccine.
Recommended
A final analysis conducted later in the month found the drug to be 95 percent effective with consistent results across "age, gender, race and ethnicity demographics." Pfizer says no serious safety concerns related to its vaccine candidate have been reported.
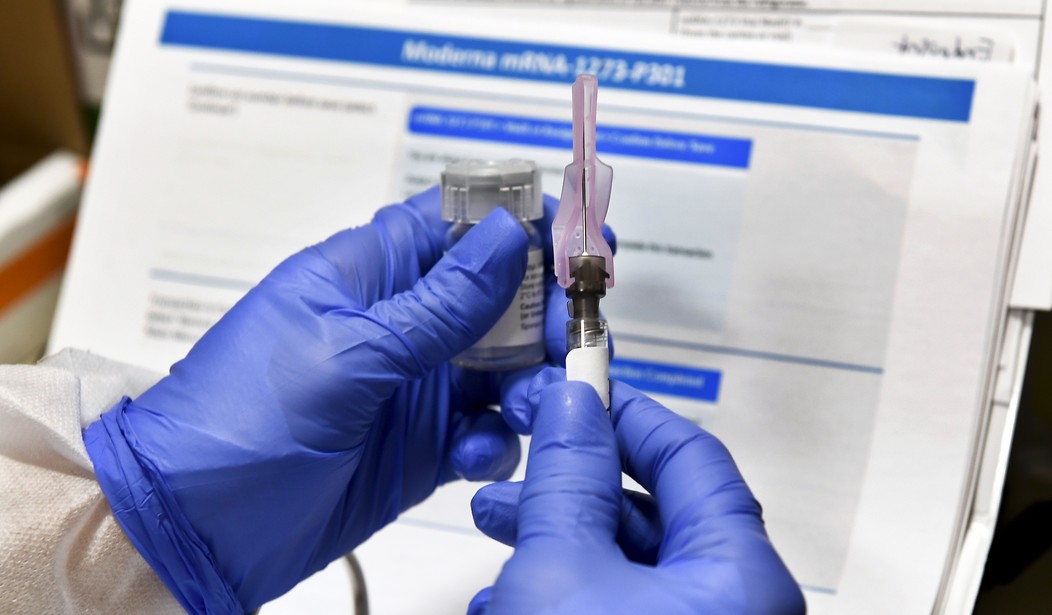
Drugmaker Moderna's vaccine candidate has proved to be over 94 percent effective in clinical trials and the drugmaker has also requested EUA from the FDA.
According to the Centers for Disease Control and Prevention, healthcare workers and residents of long-term care facilities should be prioritized for vaccination. Individuals over the age of 65, essential workers, and those with underlying medical conditions should be vaccinated in a second phase.























Join the conversation as a VIP Member