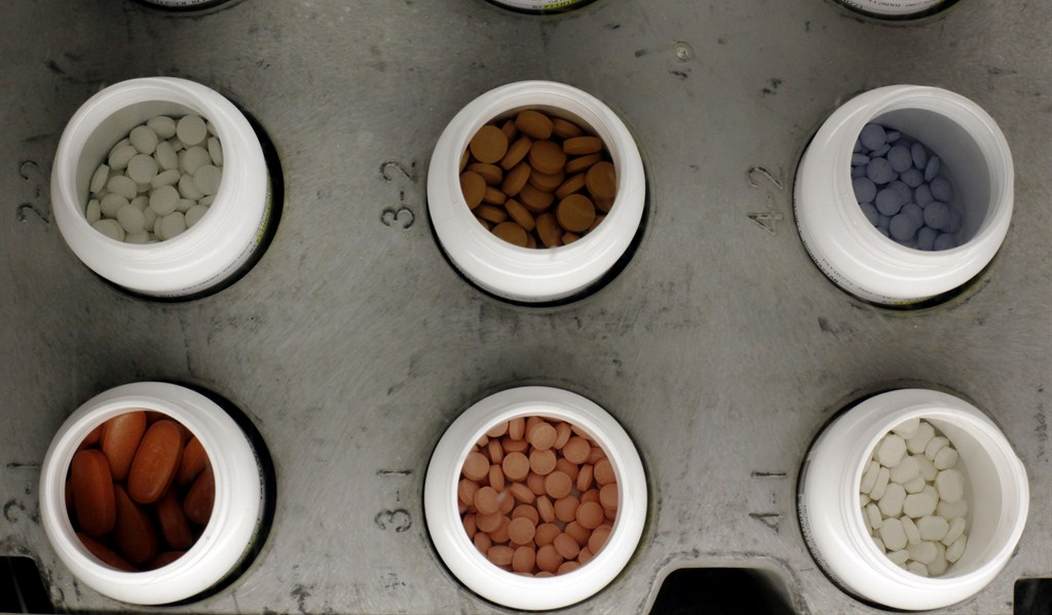
Generic drugs make up 90 percent of all prescriptions filled in this country. While many consider them nothing more than a cheaper copy of their brand name counterparts, a flurry of recent recalls have called into question the safety and quality of some of these low-cost alternative drugs.
A recent NBC News piece with the provocative title, “Tainted drugs: Ex -FDA inspector warns of dangers in U.S. meds made in China, India,” highlights the quality issues that have arisen at several overseas facilities where generic drugs are manufactured. The report highlights a major recall of the generic blood pressure drug Valsartan, found to have been contaminated with a substance used in the production of rocket fuel, a recognized probable human carcinogen. Due to these safety concerns over half of the valsartan available for sale in the United States was ultimately pulled from store shelves in August 2018.
One of the former Food and Drug Administration (FDA) inspectors interviewed for the piece was Massoud Motamed, a Texas native with a Ph.D. in biochemistry, who worked mostly abroad inspecting foreign manufacturing facilities. His assignment abroad along with those of other FDA inspectors reflects the new reality in the pharmaceutical industry. According to FDA data, approximately 85 percent of the facilities manufacturing the ingredients in American drugs are located overseas, including many from China and India where production costs are low, regulation is lax and local government oversight is less stringent.
Recommended
The issues related to tainted Valsartan are unfortunately not isolated. Since the summer of 2018, drug companies have announced a total of 45 recalls of generic blood pressure medications alone, including some versions of Valsartan as well as two other blood pressure drugs, Losartan and Irbesartan. The raw ingredients in these drugs all came from overseas manufacturing sites.
With so much focus in the U.S. on cost containment, a push toward generic drugs has been strong and sustained over the last several years. As a result, the vast majority of ingredients used in generic American pharmaceuticals are manufactured abroad, where the ability to inspect for quality is much more limited. FDA inspectors abroad typically count on drug manufacturers to identify and report problems through company testing but there is direct evidence that at least one plant engaged in unethical activity prior to an FDA visit and it is reasonable to conclude that this may not be an isolated incident.
Furthermore, though the number of generic drugs approved by the FDA increased 94 percent since fiscal year 2014, the number of FDA surveillance inspections done globally – to ensure drug-making plants meet U.S. standards – dropped 11 percent in fiscal year 2018.
Doctors have also expressed concern over the safety and effectiveness of the drugs that they prescribe. Dr. Randall Zusman, a cardiologist at Massachusetts General Hospital Heart Center, stated, “It’s hard to know what to prescribe patients. You want to assume it’s safe and effective. You don’t want to feel like you are prescribing something that causes harm.” In addition to doctors, it is vital that patients not lose confidence in the safety and effectiveness of the drugs being dispensed to them.
Drug safety laws in America date back to the Pure Food and Drug Act of 1906, the first of a series of significant consumer protection laws enacted by Congress in the 20th century. The main purpose of the Act was to ban foreign and interstate traffic in adulterated or mislabeled food and drug products. Statute directed the U.S. Bureau of Chemistry to inspect products and refer offenders to prosecutors. The act also required that active ingredients be placed on the label of a drug’s packaging and that drugs could not fall below purity levels established by the United States Pharmacopeia or the National Formulary.
While cost is a major factor in healthcare, it is not the most important factor. As Congress acknowledged over 100 years ago, quality is paramount when it comes to drugs intended to regulate vital medical functions.
We all grew up with the saying, “You get what you pay for.” Unfortunately, when it comes to pharmaceuticals, our national health policy seems to prioritize cost over quality and generics over their branded counterparts. Since such a high percentage of generics are manufactured in overseas facilities with limited oversight and since insurance companies substitute generics for branded drugs when available, the promise of the Pure Food and Drug Act of 1906 and is successors seems to be moving further away from us as our national policy in effect values low cost over quality. A policy course correction is in order before people get hurt.























Join the conversation as a VIP Member