By now, you're aware of the Trump administration's worthy "Operation Warp Speed" project -- which we told you about weeks ago, and which the president formally unveiled late last week. In case you missed the rollout, it featured prominent members of the task force and a surge of optimism about the government's ability to partner with private industry to develop a coronavirus vaccine in record time. Here's a clip from Friday's Rose Garden event formally christening the dramatic push, which has been likened to a modern-day Manhattan Project:
President Trump on Friday announced "Operation Warp Speed" -- a new initiative that is aimed at developing, manufacturing and distributing a "proven" coronavirus vaccine. Trump described the administration's plan as "a massive scientific industrial and logistical endeavor unlike anything our country has seen since the Manhattan Project" of World War II, with the intent to rapidly develop and distribute a vaccine with help from the U.S. military and world-renowned doctors and scientists. "We'd love to see if we can do it prior to the end of the year," the president said. "We think we're going to have some very good results coming out very quickly."
Trump has mentioned a similar timeline in the past, causing some critics to charge that he's over-promising. I agree that it's politically risky for the president or his administration to raise expectations on an imminent vaccine, but as I explained in this piece, it's possible that they're on fairly solid ground substantively, and also have little choice politically. Right now, it looks like the upcoming election could very well wind up being a referendum on Trump's handling of the dual public health and economic crises gripping the country. A reliable vaccine by the fall could represent a bona fide game-changer for a president who may need one badly:
Recommended
“I think it still boils down to a referendum on the president. They’ll beat up on Biden and they’ll raise some concerns,” said the former two-term Republican governor of Wisconsin, who lost his seat in 2018. But in the end, if people felt good about their health and the state of the economy, Trump will probably carry Wisconsin. If not, Walker said, “it’s much more difficult” for the president...Interviews with nearly a dozen former governors, members of Congress, and other current and former party leaders revealed widespread apprehension about Trump’s standing six months out from the election. Many fret that Trump’s hopes are now hitched to the pandemic; others point to demographic changes in once-reliably red states and to the challenge of running against a hard-to-define Democratic opponent who appeals to a wide swath of voters. The concerns give voice to an assortment of recent battleground state polling showing Trump struggling against Biden.
Talking up the prospect of a vaccine in an unrealistic way may punt political problems into the future while selling hope, but if the fall rolls around with no vaccine in sight, an already-precarious electoral situation could grow dire. But there's growing evidence that Trump and his team aren't simply peddling aspirational feel-goodery. A vaccine candidate out of Oxford, and tested on monkeys at a lab in the United States, generated promising outcomes back in April. And now there's this highly encouraging news from a leading American candidate:
Moderna (the leading coronavirus vaccine candidate in the US) released some early data on its vaccine trial.
— Alicia Smith (@Alicia_Smith19) May 18, 2020
Antibodies were produces in all 45 patients who received the vaccine. https://t.co/OvdUJEmjUG
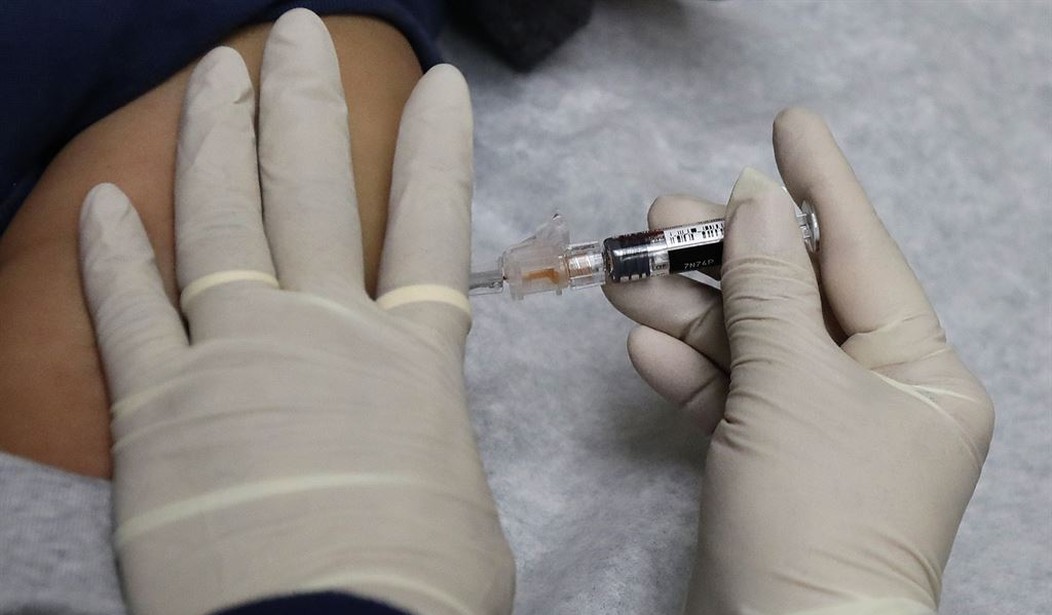
The first coronavirus vaccine to be tested in people appears to be safe and able to stimulate an immune response against the virus, its manufacturer, Moderna announced on Monday. The findings are based on results from the first eight people who each received two doses of the vaccine, starting in March. Those people, healthy volunteers, made antibodies that were then tested in human cells in the lab, and were able to stop the virus from replicating — the key requirement for an effective vaccine. The levels of those so-called neutralizing antibodies matched the levels found in patients who had recovered after contracting the virus in the community. The company has said that it is proceeding on an accelerated timetable, with the second phase involving 600 people to begin soon, and a third phase to begin in July involving thousands of healthy people. The Food and Drug Administration gave Moderna the go-ahead for the second phase earlier this month.
This is excellent news, full stop. Sure, we should remain cautious; it's a tiny sampling, after all, and perhaps problems will arise in the larger test pools. Things could go sideways. But if the second and third rounds are even in the same ballpark of success, we'd be looking at a monumental breakthrough and achievement. More details, via The Wall Street Journal:
For some participants ages 18 to 55, varying doses of the vaccine increased immune responses, including boosting certain antibodies to levels at or above those seen in blood samples from people who have recovered from Covid-19, Moderna said. The responses included both binding antibodies, which attach to viruses but don’t prevent infection, as well as neutralizing antibodies, which do block infection...The phase 1 study data reported Monday were from portions of the 45 people ages 18 to 55 who received three different dose levels of the vaccine. An additional 60 people over age 55 are being enrolled in the study. Moderna said one participant experienced redness around the injection site, and three subjects receiving the highest dose had “systemic symptoms.” Moderna described the adverse events as transient and self-resolving...Moderna also said Monday a study of the vaccine in mice showed that it prevented viral replication in the animals’ lungs after they were exposed to a modified version of the coronavirus.
Broad effectiveness, minimal and fleeting side effects, and as Allahpundit notes, there's at least some evidence that this innovative type of vaccine could be cheaply mass produced: "mRNA vaccines have the potential for rapid, inexpensive and scalable manufacturing, mainly owing to the high yields of in vitro transcription reactions." Former FDA chief Dr. Scott Gottlieb sounds heartened:
Encouraging news today on Moderna vaccine. Technology will eventually let us greatly reduce the covid threat and reclaim normal times. Early data shows it generates robust immune reaction, and its dose dependent. Getting dose right is key. Moderna now testing a new 50mcg dose 1/x
— Scott Gottlieb, MD (@ScottGottliebMD) May 18, 2020
He adds that some data indicate that some of the vaccine candidates may not prevent or defeat the virus altogether, but like a flu shot, they could at least mitigate COVID's worst symptoms. Raise your hand if you'd take such a drug in a heartbeat, if it was clinically proven to be safe. "The good news," Gottlieb says, is that "there's now accumulating evidence across multiple products that we should get a vaccine." As we wait for additional trial phases to play out, I'll leave you with this:
In most states the number of cases are either declining, or flat. Only a handful of states are showing expanding cases based on common models. We expect cases to grow as mitigation is withdrawn; we need to watch closely to make sure the increases are manageable and containable. pic.twitter.com/VKN6tRDf7h
— Scott Gottlieb, MD (@ScottGottliebMD) May 17, 2020
We now have "evidence of an epidemic that slowed dramatically," he writes. "While [the virus] is still expanding, it's doing so at much slower pace and hopefully there'll be a seasonal effect in the Summer that slows it further." Fingers crossed, prayers up, etc.
Editor's Note: Want to support Townhall so we can keep fighting the ChiComs and tell the truth about China and the virus they unleashed on the world? Join Townhall VIP and use the promo code WUHAN to get 25% off VIP membership!
























Join the conversation as a VIP Member